In short: rust is iron oxide — it only dissolves with an acid (citric, lemon, oxalic). The absolute rule: never use bleach, which further oxidises the iron and permanently sets the stain. Citric acid powder is the most reliable solution. Oxalic acid salt for stubborn cases (with precautions).
At a glance
NEVER bleach — it oxidises the iron and sets the stain. Mistake number 1.
Citric acid — the most reliable treatment. 1 tbsp in half a glass of warm water, 15-30 min.
Lemon + salt in the sun — traditional method, effective on white cotton.
Oxalic acid salt for stubborn cases — powerful but toxic, gloves mandatory.
Never hot water before treatment — heat sets iron oxide deposits.
Why rust stains (and why it is so unusual)
Rust is iron oxide (Fe2O3) — a mineral compound totally different from organic stains like coffee, red wine or blood. Where tannins bind to fibres through fragile hydrogen bonds, iron oxide forms mineral deposits that physically embed in the fabric structure.
Rust chemistry on textiles
When an iron object oxidises on contact with moisture, it produces iron (III) oxide — Fe2O3 — an orange-brown powder insoluble in water. On contact with damp fabric, the oxide microparticles lodge between fibres and cling through electrostatic and mechanical forces.
To remove rust, you must dissolve the iron oxide. Only an acid can do this, by converting Fe2O3 (insoluble) into an iron salt (soluble), which rinses away. Citric acid↗ is particularly effective because it works by chelation: its molecules “trap” the released iron ions, making them soluble and preventing re-deposition on the fabric.
Why alkaline products do not work
Standard detergents, [sodium percarbonate](/blog/percarbonate↗-de-soude-linge/), baking soda and Marseille soap↗ are alkaline (pH > 7). In an alkaline environment, iron forms even more insoluble iron hydroxides. A machine wash without prior treatment can therefore make things worse — the stain sets further instead of coming out.
Why bleach is catastrophic on rust
Bleach (sodium hypochlorite) is an oxidiser. On rust, it does exactly the opposite of what is needed: it further oxidises the iron, fixes it more deeply in the fibres and turns the orange stain into a permanent dark brown mark. This is the most common mistake — and often irreversible.
The citric acid + salt method: detailed step by step
Citric acid powder (available at hardware stores and supermarkets, 3-5 euros/500 g) is the most reliable treatment. It dissolves iron oxide by chelation — citric acid molecules encapsulate iron ions and make them soluble.
Dissolve 1 tbsp in half a glass of warm water — warm water (not hot) speeds up powder dissolution without setting the rust.
Add a pinch of fine salt — salt acts as a mild abrasive that helps dislodge oxide particles from the fibre surface.
Apply to the stain — soak the fabric or immerse the stained area. You can also place a soaked cotton pad directly on the stain to maintain contact.
Leave for 15-30 minutes — the stain gradually fades as the iron dissolves. If it does not fade after 15 minutes, the concentration may be too low — add more citric acid.
Rub gently and rinse — thoroughly with cold water to flush dissolved iron salts.
Repeat if necessary — old stains may need 2-3 successive applications. Each one dissolves an additional layer.
Dosage for large stains: if rust covers a large area (laundry dried on a rusty wire, for example), soak the entire garment in a basin with 3-4 tablespoons of citric acid per 5 litres of warm water. Leave for 30-60 minutes.
The lemon + salt + sun method
This is the traditional method, effective on light to moderate stains on white cotton. The sun plays an active role in the process.
- Squeeze lemon juice directly on the stain.
- Sprinkle fine salt on top.
- Expose to direct sunlight for 1-2 hours — UV accelerates the photochemical reaction. The citric acid in lemon dissolves the iron oxide, while UV triggers a photoreduction reaction that helps whiten residues.
- Rinse with cold water and machine wash.
Limits: not very effective on old stains or coloured textiles (lemon can lighten dyes). In overcast weather, the method works but takes longer (3-4 hours).
Stubborn cases: oxalic acid salt
Oxalic acid salt (oxalic acid) is the most powerful reducer for rust. It is significantly more effective than citric acid, but also more toxic.
Mandatory precautions with oxalic acid salt
Oxalic acid is toxic if ingested and irritating. Wear gloves, do not inhale the powder, work in a ventilated room, rinse the textile thoroughly after use. Keep out of reach of children. Never mix with bleach — the reaction can produce irritating gases.
- Dissolve 1 teaspoon in 250 ml of hot water.
- Apply to the stain, leave for 10-15 minutes maximum.
- Rinse thoroughly (3-4 rinses) to remove all residue.
Oxalic acid salt works by reduction: it converts Fe3+ (insoluble, orange) to Fe2+ (soluble, colourless). This is the most direct and fastest mechanism for dissolving rust.
What does NOT work on rust
Certain methods popular for other stains are counterproductive on rust. Here is why:
Bleach
An oxidiser that worsens iron oxidation. Turns the orange stain into a permanent dark brown. The most serious and common mistake.
Hot water / tumble dryer
Heat accelerates oxidation and sets iron oxide deposits in the fibres. Never wash hot before eliminating the rust.
Sodium percarbonate
An alkaline oxidiser. It oxidises iron (like bleach, but milder) and the alkaline environment forms insoluble iron hydroxides. Percarbonate is excellent on wine or coffee, not on rust.
Standard detergent alone
Detergents are alkaline (pH 9-11). A machine wash without prior acid treatment can set the rust further. Always treat before washing.
Marseille soap
Alkaline (pH ~9-10), it does not dissolve iron oxide. It may shift surface particles but does not chemically eliminate them.
How to prevent rust stains
Prevention is more effective than treatment. A few simple steps eliminate the most common causes:
- Empty pockets before washing — a single forgotten pin, screw or paperclip can leave orange traces on the entire cycle.
- Check the clothesline and drying rack — rusty metal wires transfer rust as soon as wet laundry touches them. Replace them or use plastic pegs.
- After a water cut: run the tap for 1-2 minutes before starting a machine. Old pipes release rust particles after an interruption.
- Storage: do not place damp laundry on metal surfaces (shelving, cast iron radiator, garden chair).
- Inspect the machine drum: old washing machines can develop rust on the drum or seals. Visually inspect and run a white cloth over metal surfaces. If the cloth comes out orange, clean or have repaired. See our guide to cleaning a washing machine.
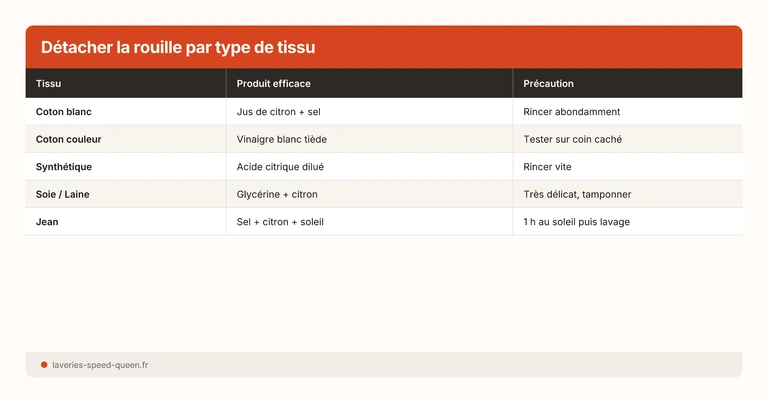
By fabric: adapting the treatment
White cotton
The most treatable. Citric acid or oxalic acid salt without risk of discolouration. The lemon + salt + sun method works very well. After treatment, you can whiten yellowed laundry if traces remain.
Coloured cotton
Citric acid is safe. Avoid prolonged oxalic acid salt and lemon in the sun — risk of lightening the dye. Test on a hem.
Synthetic
Good results with citric acid. Polyester retains less surface rust than cotton — particles cling less to non-polar synthetic fibres.
Silk
Strong acids are risky on protein fibres. Dab with very diluted lemon (1/3 lemon, 2/3 water) and consult a dry cleaner if the stain persists. Never rub silk.
Wool
Same caution as silk — protein fibre sensitive to acids. Diluted lemon, short contact time (10 min max). For valuable wool garments, dry cleaning is recommended. See our delicate textiles guide.
Linen
Cellulose fibre, resilient like cotton. Citric acid works very well. Linen absorbs heavily — use a generous amount of solution.
Mistakes to avoid
- Using bleach — the most serious mistake. Bleach oxidises iron and permanently sets the stain. Often irreversible.
- Washing hot before treatment — heat sets iron oxide deposits in the fibres.
- Using an alkaline product — standard detergents and percarbonate are alkaline. Rust requires an acid. Treat before washing.
- Skipping gloves with oxalic acid salt — oxalic acid is irritating to skin and toxic if ingested.
- Not rinsing enough — concentrated citric acid or oxalic acid residues can weaken fibres if left too long.
- Mixing oxalic acid salt and bleach — the chemical reaction produces irritating gases. Never mix these two products.
- Treating dry — citric acid powder directly on dry fabric can create rings. Always dissolve in water first.
As an Amazon Partner, we earn a small commission on purchases made through the affiliate links in this article — at no extra cost to you. This helps us maintain this site and produce free guides.
Our laundromats in Blagnac, Croix-Daurade and Montaudran have professional machines with detergent included. The higher water volume (50-60 litres) helps thoroughly rinse anti-rust treatment residues. Payment CB sans contact ou espèces. See our prices.
Sources and references
- Stain removal: solutions for all stains
- Whitening yellowed laundry (related traces)
- Wash temperature guide
- White vinegar and laundry: uses and limits
- Cleaning your washing machine
- Delicate textiles care
- Chelation chemistry — citric acid forms stable complexes with Fe3+ ions, making iron oxide soluble
- Photoreduction of iron-citrate complexes under UV — natural whitening mechanism documented in photochemistry